California Life Sciences Association (CLSA) Releases New Infographic Detailing Improved FDA Drug Review Performance
May 1, 2016
By California Life Sciences Association
The FDA plays a critical role in the drug review and approval process, with twin goals of employing rigorous scientific and safety standards to ensure new medicines get to patients as quickly as possible.
On March 29, the California Life Sciences Association (CLSA) released a new infographic report on the U.S. Food and Drug Administration’s (FDA) drug review and approval times over the last 15 years. Produced in partnership with The Boston Consulting Group, the infographic, titled, “Tracking FDA Drug Review Performance,” shows that the FDA has accelerated review times, descending from an average of 21 months in 2009 to 10 months in 2015.
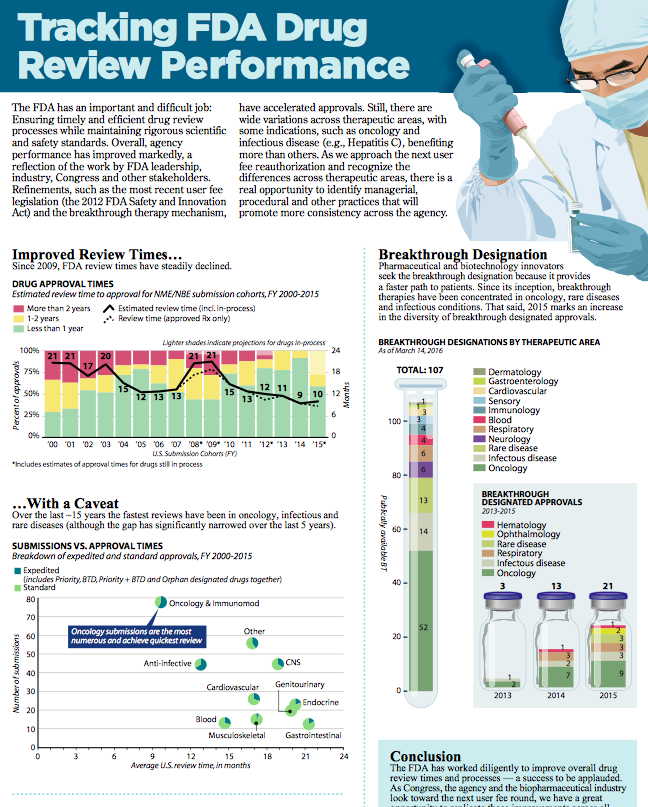
Key facts and infographic highlights:
- Since 2009, drug review times have steadily improved, with particularly fast review times seen in the therapeutic areas of oncology, infectious, and rare diseases.
- Despite consistent decreases in average approval times, endocrine, gastrointestinal and CNS therapies have lagged behind in recent years.
- 2015 marked an increase in the diversity of breakthrough approvals since the inception of the ‘Breakthrough Therapy Designation.’
The infographic report has garnered significant press coverage, in outlets including: Newsweek, STAT News, InsideHealthPolicy, MedCityNews, Regulatory Focus, Life Science Daily, SmartBrief and more.
View the full infographic and download at http://www.califesciences.org/2016FDADrugReport.

Stay Connected
Sign up for updates straight to your inbox.
