Economies of scale are the efficiencies unlocked by large numbers. I’ve come to realize that the efficiencies of scale can emerge at the opposite extreme of rare—especially ultra-rare—genetic disease communities, which can number as few as dozens to thousands of members, globally and at any given moment in time. I call it economies of rare.
Economies of scale are the efficiencies unlocked by large numbers. I’ve come to realize that the efficiencies of scale can emerge at the opposite extreme of rare—especially ultra-rare—genetic disease communities, which can number as few as dozens to thousands of members, globally and at any given moment in time. I call it economies of rare.
Instead of savings coming from increased production, savings come from increased elimination. Whether it’s the costs of coordinating and commercializing research, community management and patient engagement, or clinical trials themselves, rare disease communities—where everybody knows somebody willing to pitch in—find a bypass around every roadblock and trim all the fat from the traditional pharma cost structure. Opportunities for unimaginable progress spontaneously emerge from collaborations, cross-pollination and in-kind contributions.
Turning rare into scale
Over the last five years, I’ve seen the same heart-rending and hope-inspiring sequence of events unfold when rare disease families and their social networks mobilize. In the flash of a diagnosis, random strangers from around the world connected only by the Internet and a shared genetic lesion realize they belong to the same long-lost monogenic tribe.
Like the groups of our ancestors that risked life and limb to venture across land bridge or open ocean, small numbers of patients, advocates, scientists and stakeholders hastily set out to cross an uncharted and unforgiving continent of constant caregiving, coalition building, fundraising, basic science, and drug discovery and preclinical development, culminating in clinical trials and the promised land of FDA approval.
How do they take that first step into the unknown? The answer is teamwork.
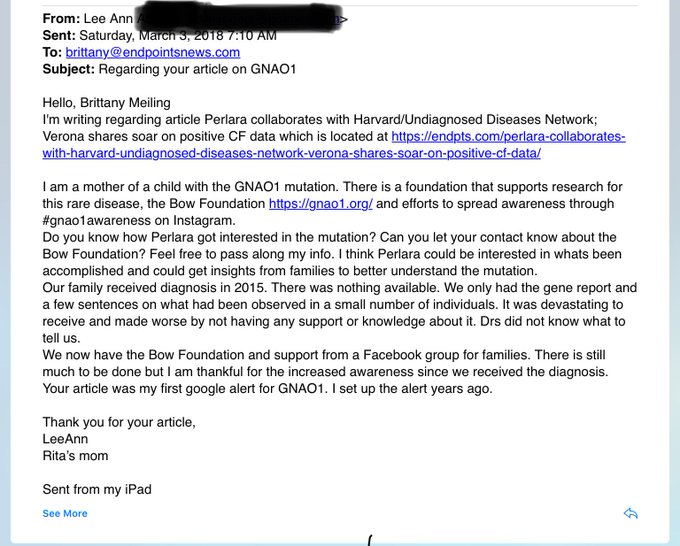
Clinical exome/genome sequencing and social media started to converge 8-10 years ago, the first step toward economies of rare. Nowadays, a tweet or a Facebook group can catalyze a chain of events leading to the formation (or reactivation) of rare disease communities. For example, consider this email from a parent and advocate for GNAO1. She hadn’t received a single Google alert for GNAO1 since 2015 until our press release last week.

Thanks to an Internet connection, the costs of connecting have essentially dropped to zero for anyone determined enough to search for their tribe. That means rare disease communities coalesce faster, or in many cases, for the very first time.
The patient as the hub
What happens after the community first gels? They start executing on the plan. The plan is always the same: find a cure. Some members of the community move faster or slower, tilt more in one strategic direction or another, or have more or fewer resources at their disposal, but they’re all still guided by the same compass. In contrast to the blockbuster playbook, where companies and researchers recruit patient communities, rare disease communities flip the script by recruiting researchers and companies into their orbit.
When the Orphan Drug Act passed 35 years ago with the noblest of intentions, it was not expected that rare disease communities would actually sit in the driver’s seat from diagnosis to the clinic. Leave the R&D to the dispassionate, or so went conventional wisdom. The patient focus was supposed to on caregiving or educational awareness or policy. As important as those goals are, they mean nothing without new research. We can only science our way to cures.
Rare disease foundations are increasingly focusing their mission and their dollars on new research at all stages. They usually start with the one or a few scientists, almost all academics, who have studied their gene the longest, as a labor of love. Often these scientists cloned the gene 15 or 20 years ago. However, there may not be many labs working on the gene today. It’s critical to recruit fresh blood into research consortia that otherwise gravitate toward groupthink or fail to diversify therapeutic strategies.
Rare disease foundations today provide not only grant support and co-investment but also a forum for precompetitive exchange of unpublished data. The best ones achieve a balance between basic scientists, translational scientists, physician scientists, and clinicians on SABs, as well as representatives from industry spanning early-stage biotech to mature biopharma companies.
Patient equity, urgent mission
The institutional investors who usually fund new biopharma companies are writing larger and larger Series A checks to a narrower and narrower segment of managements teams, usually repeat CEOs or SVPs and ex-C-suite from large established companies with long track records. Economies of rare are just now at an inflection point where new financing and business models allow rare disease foundations to advance de-risked discovery-stage and preclinical-stage data to the commercial level as therapeutics platform NewCos.
The business case is obvious to anyone who spends time in the rare disease community: insights into rare diseases shed light on genetically-related common diseases. The most popular example of this phenomenon is the gene PCSK9, a disease modifier where mutations that activate PCSK9 cause hypercholesterolemia and premature cardiovascular disease, while mutations that inactivate PCSK9 cause hypocholesterolemia and are cardio-protective. I believe human genetics will show that PCSK9 is the rule rather than the exception. We need to look no further than the Perlara PerlQuest pipeline for several other examples of the rare-to-common thesis.
Take NGLY1. NGLY1 Deficiency is our first PerlQuest disease, and the Grace Science Foundation is our first PerlQuest partner. Last year, NGLY1 biology was connected to cancer, such that enhancers of NGLY1 Deficiency might make for potential cancer drugs, while suppressors of NGLY1 Deficiency would of course make for potential therapies for NGLY1 Deficiency. Now there’s a venture-backed NGLY1 therapeutics platform company called Grace Science, LLCthat is ready to take the baton once the discoveries made by GSF are ready for commercialization. Will every rare disease foundation one day create a companion for-profit disease gene platform company leveraging the rare-to-common thesis?
NGLY1 Deficiency is lucky compared to other ultra-rare disease communities in that it has more than one consortium in the race for a cure. NGLY1.org, NCATS and Retrophin joined forces late last year in a public-private-patient advocacy group/PAG partnership. So there are at least two paths available for rare disease foundations to choose from as they transition from discoveries to drug candidates and beyond, and thanks to economies of rare, more paths will surely open where none were before.
Ethan Perlstein is CEO of Perlara, the first biotech public benefit corporation (bioPBC) that discovers definitive treatments for rare diseases and learns how genes work across diseases to map connections to common ailments. Perlara embarks on journeys of scientific discovery called PerlQuests with families, patient organizations, and BioPharma and clinical partners. This article originally appeared in Perlara’s blog, “The Ark“

Stay Connected
Sign up for updates straight to your inbox.